It's All About Balance Fertile soil is a mixture of well-balanced minerals, high organic matter, humus, humic, fulvic and carbonic acids, good aeration and bountiful microbial life. The biology or life in the soil is at its healthiest when the nutrients are plentiful and balanced, and there is sufficient oxygen and water. The top soil region is the most vital portion of the soil profile; holding about 70% of the life and 70% of the organic matter. In a typical soil, below 6 inches plant roots are feeding on mostly soluble nutrients since the micro-organisms are not able to thrive due to insufficient oxygen levels. Many minerals are tightly bound to the subsoil colloidal particles are only made available to plants through complex soil interactions with organic acids leaching downward from the topsoil. It is critical to maintain the organic matter content in soils for them to remain balanced and healthy. Reducing soil compaction is a major primary factor in maintaining nutrient availability. Simply put; higher levels of oxygen at deeper soil depths increase the total quantity of micro-organisms like bacteria, fungi, actinomycetes, algae, nematodes and protozoa. These microbes contribute directly to the release of soil nutrients to the plant. Some species of specialized symbiotic mycorrhizae can tolerate low oxygen levels and can colonize roots much deeper than other species of beneficial microbes; providing deeper sources of nutrients and providing root protection against pathogens. There are many very complex symbiotic relationships going on between plant roots, organic matter, colloidal soil particles and micro-organisms that support overall plant growth. It is important to note that soil that is worked too wet destroys air and water soil pore space, destroying the oxygen-rich environment that microbes need to thrive. Soil that is worked too dry creates similar problems. Tending soil for optimum production means adding minerals and organic matter every year when soil conditions are optimum. Well balanced fertile soil makes for higher crop yields, higher quality, with less disease and insect pressure. Soil testing may indicate all nutrients and minerals may be present but these may not be available to the plants if the soil is poorly maintained. Complete soil testing Many hop growers do not realize the value of accurate and timely testing of their soils in their hopyards. The goal of all soil testing is to inform the grower of what nutrients are available at the beginning of the growing season; middle of the season, and what is left at the end of the season. These tests help identify nutritional problems before they cause issues and help determine what needs to be amended. It is strongly advised to do soil/foliage testing three times each season. A complete soil test includes: Growers should become familiar with how to interpret the soil and foliar test results. Knowing why, when, and how to complete a field test is equally important to getting accurate results. Here's the basics:
Taking a Soil Sample © willington crop services © willington crop services The aerobic topsoil zone is usually only 6-7” deep and should be the primary zone tested for most crops. Hopyard soil samples should be taken from in the hop row; within 18 to 24 inches of the hop’s crown structure, where most nutrient depletion occurs. Perennial crops like hops may need to be sampled at greater depth to determine toxicity issues such as sodium, boron, carbonates, or residual herbicides. If the soil has been mechanically deep-tilled with a subsoil implement or plowed, soil sampling could go to 20” plus. Using a tube type soil probe or a shovel, take a minimum of five probes in different zones of the area being tested. Scrape away any surface organic material or you will get an abnormally high reading. Do not combine probes from areas that are not uniform to the sample desired; for example when observing rocky, clayey, silty, flooded conditions; or when noting differences in crop or weed growth occurs. Sample these areas separately but do not combine them with the “normal” sample. Mix the representative 5 or 6 soil probe samples together to create a single sample. Mix deep samples separately from shallow samples. Combined it should be about 1½ to 2 cups minimum of mixed soil. Do not touch or mix the soil samples with your hand. Don’t use a rusty shovel. Place sample in a container approved by the soil testing lab of your choice.
The highest rates of soil fertility are seen when the testing done in May and June; the lowest in the winter. To get consistent comparative results, test soil at the same time of the year. This allows comparisons of the annual soil tests to spot changes in the soil fertility from season to season. Reading a Soil Test There are many labs around the country that give soil test results. Every lab uses different testing methods so the comparative numbers may not be the same lab to lab. It is best to select one lab consistently to track annual testing results. The categories below are all part of a complete soil test. (Note: To covert pounds per acre to pounds per 100 square feet divide by 440.) Organic Matter (O.M.) Increasing organic matter levels will help with the soil texture, structure, drainage, aeration, water holding capacity and availability, nutrient availability, root development and dramatically improve soil biology. Organic matter (humus) holds three times more nutrients than most clay soil types and up to 5 times as much available water. 2% or less organic matter is considered poor. Over 4% O.M. to 10% is ideal. Above 10% organic matter can often inhibit micro-nutrient uptake, and if composed primarily of woody materials will greatly reduce nitrogen availability. (Microbes breaking down wood cellulose use large amounts of nitrogen.) Most crop soils benefit from adding organic matter every year; especially if tilled or intensively farmed. Adding composts, manures, cover crops, and other organic mulches are the best choices for increasing organic matter. Side Note: Working overly wet soils destroys organic matter and adds to soil compaction. Adding fresh non-composted organic matter such as raw wood chips high in cellulose will require additional nitrogen to compensate for the amount used by microbes breaking down the cellulose fibers. Phosphorus (P) Phosphorus is an important macro nutrient used in the storage and transfer of energy in the plant. It is essential in every metabolic growth process, protein synthesis, sugar development, and in nitrogen fixation. It is crucial for new root development. Optimum phosphorus levels are needed for rapid seedling and root growth, winter hardiness, disease resistance, efficient water use, early maturity, and maximum yield. Phosphorus needs to be placed and incorporated where it will be used; as it is less mobile in the soil than any other nutrient. It does not leach down in the soil profile like the macro nutrients nitrogen and potassium. Some cover crops can move phosphorus to deeper areas of the soil, where it will become available for other crops only after the cover crop roots decay. This is why cover crops should be plowed down and incorporated; rather than being left as a perennial row cover. Phosphorus becomes immobilized at low pH by large concentrations of aluminum, zinc and iron, and at high pH by too much calcium. Soils with high levels of Al, Zn, or Fe may need additional P applications to counter balance and provide adequate levels for optimal plant growth. Soft rock phosphate is the fastest working phosphate. 300#/acre is the normal recommended minimum application. Up to 2000#/acre of soft rock phosphate is often incorporated in severely deficient soil types. Phosphites are not the same as phosphates! (Read More) A complete soil test may contain two phosphorus tests:
Potassium (K) 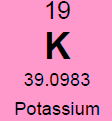 Potassium is a macro nutrient that regulates the pace of plant metabolic activities. It is essential for photosynthesis and protein synthesis as well as carbohydrate transport and storage. It promotes root sugar reserves, winter hardiness, cell development, strong cellular walls and stems. Potassium improves water use efficiency, increases yield, improves crop quality, and reduces incidence of disease. Potassium is mobile within a plant and can translocate from older leaves to newer growth. Soluble potassium is easily leached from soil profiles and needs to be replaced annually; like nitrogen. Most soils have less than 1% of the existing potassium available due to insufficient microbial activity and organic matter content. There are about 30,000 to 50,000 lbs. per acre of potassium in an average soil, but most of this is not plant available until microbial activity releases it. It is possible to release small amounts of potassium over time by increasing microbial activity with compost, compost tea and cover crops. It is the second most probable nutrient to be deficient after nitrogen. Applications of granular sulfate of potash (50% K2SO4) are timed in early fall (Read More) and spring to perennial crops. Additional soluble grade potassium fertilizer may be added to irrigation during the growing season, or foliar fed if severely deficient. Avoid winter and late fall applications after plants have gone dormant. Excess potassium will not create toxicities, but will block uptake of other nutrients; so single large applications are not as favorable as multiple applications throughout the season. Too much potassium ties up boron, calcium and manganese. 2% Cation saturation potassium is the minimum and 5% to 7% is more optimal. Magnesium (Mg) 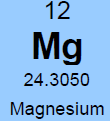 Magnesium is an essential micronutrient found in the chlorophyll of green plants. It is also necessary for metabolic processes and in every operation involving phosphorus. Magnesium levels have important interactions with calcium, sulfur, and nitrogen. The ratio of magnesium to calcium should be around one to six. Excess magnesium will reduce potassium availability. Having a soil with too much magnesium will take more nitrogen because the excess magnesium makes the soil colloids bind too tightly. Excess magnesium is what makes most clay type soils “tight”, restricting air and water availability, water drainage, root development and restricting microbial activity and organic matter decay. Applications of garden gypsum are often recommended for clay-type soils with elevated levels of Mg to loosen the soils. Higher levels of magnesium in a sandy soil will help to tighten loose sand. For sandy soil the optimum level would be 16% to 20% and for clay soils closer to 12%. Mg availability is also closely tied to soil pH; so it is important to monitor the soil pH. Note: Two pounds of sulfur will leach out one pound of magnesium when there is at least 60% calcium saturation. Calcium (Ca) Calcium is the building block of cell division with strong cell walls and membranes; it also controls water movement in and out of plant cells and reacts with plant waste products and neutralizes toxic materials. Calcium is locked into a plants cellular structure, and thus is immobile once it is utilized by the plant. Calcium activates many of the soil microbial enzyme systems; it improves microbial activity and it enhances root uptake of other nutrients. Calcium is critical for balancing excess nitrogen and for disease suppression. Having the correct amount of calcium in the soil will require less nitrogen for optimal plant growth. Calcium will help loosen compacted soil and make more nitrogen available. Too much calcium can tie up all other nutrients especially magnesium, potassium, boron, zinc and copper. Gypsum and lime are two common sources of calcium. Application of sulfur neutralizes excess calcium. Calcium cation (+) saturation levels need to be over 60% before adding additional gypsum (calcium sulfate) to lower excess magnesium levels; otherwise the sulfur in the gypsum will neutralize the calcium first. Add sufficient limestone first to raise calcium levels to 60%, and then add enough gypsum to raise calcium levels to 68%. One third of applied calcium from lime / gypsum will become available the first year and it takes 3 years to be completely utilized. Solution grade limestone will quickly become 100% available within 1-3 months. Limestone applied to the surface of the soil will work its way into the soil at the rate of 1” per year if not incorporated. Calcium leaches down in soils with excess rain or irrigation. Consult with a soil specialist to determine the proper amounts to add as amendments to avoid raising soil pH levels to undesirable levels. Sodium (Na) Widespread in most soil types, sodium is found in all plant material. Although it does not seem to be necessary for the growth and development of plants, it is taken up by the plant, particularly when potassium is low. Sodium seems to be able to partly substitute for potassium and can block potassium uptake if in excess levels. Excess sodium is a problem in many dry areas particularly if the irrigation water is alkaline. Sodium toxicity to plants is often observed in saline and alkali lands and unfavorable soil structures can be present due to high sodium as well. Excess sodium suppresses soil biology, root development and nutrient availability. Any time the sodium and potassium levels together are over 10% then the manganese won’t be able to get into the plant. Normal rainfall and irrigation leaches excess sodium out of soils. Note: Excess Chloride (Cl) levels are often associated with high sodium levels. Chlorides can reach toxic levels and should also be monitored if sodium levels are high. (Read More) Nitrogen (N)  Nitrogen is macro nutrient and is an essential constituent of proteins found in chlorophyll, enzymes, and hormones. It has a predominant role among the macro soil nutrients and is needed by plants in substantial amounts. It is also the most likely nutrient to be deficient. Deficiency limits crop growth and yield. It is rapidly taken up by crops, but much of the applied nitrogen is lost by volatilizing into ammonia, or by leaching away by water. Nitrogen in the soil is the most stable when found in organic humus and microbial bodies. Increased active organic matter and microbes increases the level of plant-available nitrogen. However, an excess of nitrogen can produce an imbalance in plant metabolism and produce overly soft growth; resulting in poorer plant quality and higher susceptibility to plant pathogens, increased transpiration water loss by the crop, reduced flavor and keeping qualities. Dangers of nitrogen overuse include: zinc deficiency, copper deficiency, burnt out organic humus and microbes, drives out calcium but leaves magnesium (tighter soil) and depletes sulfur. The more nitrogen that is applied in excessive amounts; the higher the replacement level of the other nutrients that were carried away by the leached nitrogen will have to be. Nitrogen and sulfur can leach out calcium. Nitrogen never leaches out magnesium, only sulfur does. The best methods to stabilize proper nitrogen levels are to use and incorporate cover crops, composts and manures before, during, and after each crop growing cycle. In hopyards, the bulk if the nitrogen (50 to 80%) should be available when seasonal hop growth is between 6 to 14 feet tall. Sulfur (S)  Sulfur is a component of several amino acids that are essential for forming plant protein. It helps develop enzymes and vitamins, promotes nodule formation on legumes, aids in seed production, is necessary for good root development, improves taste, increases protein and reduces nitrates. It is involved in the chlorophyll production process, and sulfur deficient plant tissue symptoms resemble nitrogen deficient symptoms. Sulfur is commonly deficient in all soils except where sulfur based fertilizers are applied. Without sufficient sulfur the rate of organic matter decomposition is decreased, due to a deficiency in sulfur reducing decomposing bacteria and Actinomycetes. Humus and organic matter helps hold sulfur in the soil. Sulfur feeds microbes and builds organic matter levels. Sulfur is very soluble and should be added in some form every spring to soils with low organic matter content. (Soils high in active organic matter often have adequate sulfur.) Two pounds of sulfur can leach out one pound of either calcium or magnesium. Start applications of sulfur if calcium levels are above 60% base saturation. Sulfur can also be used to leach out excess sodium and boron. If gypsum (calcium sulfate) is applied, the sulfur will leach out the excess sodium cations, and the calcium will replace it in the cation balance on the soil humus and clay colloids. Sulfur has a negative ion charge and will leach out most minerals (cations+, but not anions-) that are in excess. Every spring if no other sulfur source is being added and sulfur is deficient, add either 250#/acre of gypsum or 50#/acre of Tiger 90 soil sulfur to supply enough sulfur for the growing season. Split spring sulfur applications into two or three smaller applications, a month apart if possible. Many types of manure and some composts are a significant source of sulfur; so take their additive effect into consideration. Interesting side-note: Coal-fired power plants were found to add an average of 7 #/acre sulfur to USA crop soils. Now that many coal-fired power plants are being cleaned up or are being replaced with natural gas-fired plants, sulfur deficiencies are becoming more common place in many areas. Sulfur test levels of 20 ppm are the minimum recommended level and around 30 ppm is ideal. Zinc (Zn)  Zinc is a micro nutrient that is essential for the transformation of carbohydrates, development of new leaves, seed / fruit set and the formation of protein. Zinc should always be at a higher level than copper. (This is hard to accomplish with crops where copper is often applied as a fungicide)36% Zinc sulfate is a good form to apply. Good results can also be achieved with with zinc amino chelates. 10 lbs. of zinc sulfate per acre will raise zinc levels 1.8 ppm in a clay soil, more is needed than in a sandy soil. Don’t apply more than 40#/acre of zinc sulfate to raise levels to 7.6 ppm. Zinc is needed at a minimum of 4 ppm; 6 to 8 ppm is ideal and above 10 ppm is excessive. Copper is often above 10 PPM on soils where copper is used as a fungicide regularly. A foliar fed fertilizer containing zinc can be applied in situation where copper levels are above 10 ppm. Manganese (Mn)  Manganese is a micro nutrient that holds and sets fruits and is vital in many other plant functions. Manganese sulfate is 23-27% manganese and it is the best form for raising soil levels.25#/acre of manganese sulfate will raise levels by 3.5ppm. Add a maximum of 200#s/acre of manganese sulfate to raise levels 28ppm.Keep Mn levels at about 80% of iron levels. Manganese is needed at a minimum of 15 ppm, a 30 ppm level is ideal. Iron (Fe)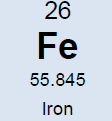 Iron is essential micro nutrient essential for the formation of chlorophyll and for photosynthesis. Iron levels always have to be higher than manganese levels; when manganese is higher than iron it will tie up the iron. Over 75% cation saturation levels of calcium will start tying up available iron. Ferrous sulfate (aka iron sulfate) is the best form to apply. (Note: Apply to foliage at low levels or it will burn the leaves and shoot tips). Ferrous sulfate will also stain concrete. Iron is rarely deficient in most soils unless the soil pH is out of range. If a plant tissue sample comes back showing a Fe deficiency; check the soil pH first before applying any iron amendments. Use an iron chelate for foliar applications. 100#/acre ferrous sulfate will raise iron levels 10.5 ppm. Don’t apply more than 400#/acre ferrous sulfate per year to raise Fe levels 44 ppm. Iron is needed at a minimum level of 20 ppm, over 40 ppm is ideal. Copper (Cu)  Copper is a micro nutrient and is an enzyme activator and is a catalyst for plant respiration. Copper and boron are disease fighters. Very high organic matter soils ties up copper; most severe copper deficiencies are on high (above 5%) organic matter soils. Excessive copper can effect phosphate, zinc and iron uptake. Growers using copper-based fungicides should be aware of this effect and monitor the other nutrients closely. Above 10 ppm CU, plenty of phosphates are needed because copper can tie up phosphorus the same way phosphorus can tie up copper. Excessive levels of nitrogen slows down the uptake of copper. Add no more than 10#/acre of 23% copper sulfate every six months to the soil; this will raise soil levels .6 ppm. A copper level in the soil of 1.5 ppm is minimal, and levels over 4 ppm are excessive. Boron (B) 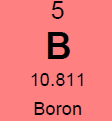 Boron is a micro nutrient that promotes maturity with increased set of flowers, fruit, seed, yield and quality. Boron is necessary for nitrogen conversion. Good boron levels make for good disease resistance. Boron is the only micro-nutrient that once corrected will still need to be applied every few years; as it leaches from soils. Boron is very potent and excess quantities can cause plant toxicities quickly. Apply boron carefully. For excessive existing boron levels, raise calcium levels to optimum first and then elevate potassium. High calcium levels can block uptake of boron, but will prevent toxic effects of excessive boron. Add no more than 10# Solubor boron per acre once a year to raise levels by .2 ppm. For fungus control keep boron levels above 1.5 ppm. (Read More) Boron levels of .8 ppm is the minimum, 1.5 ppm is ideal and 2 ppm is the maximum. The acidity and alkalinity of soil is measured as pH. For the most fertile soil most crops prefer, the cation saturation is nearly balanced and the pH will fall into a range of 6.3 – 6.8. Outside this optimal pH range many nutrients start to become unavailable and soil biology is suppressed. Decay rates and conversion of organic matter is slowed and reduced. Raising soil pH - Is not as easy as dropping some lime on it. Rates have to be calculated (see formulas below) and a soil specialist should be consulted. Different rates of lime and/or gypsum may be required (or a mix of both) to keep the levels of calcium, magnesium, and sodium balanced. Too much lime applied at once or over time can cause deficiencies of other nutrients like magnesium, potassium, boron, zinc, and copper. Gypsum is more pH neutral and will leach downward in a soil more quickly than common lime. Ground limestone only moves downward about 1 inch per year; so it is best to incorporate it into the soil with tillage equipment. Hydrated lime is very reactive compared to regular ground field lime and should be used with precaution. Soil pH is commonly adjusted lower with sulfur applications. It is important to know that sulfur applications can be very hard on fine root tips and hair feeder roots. It would be best advised to avoid heavy sulfur soil applications during periods of active root growth. Applying sulfur may be best done post-harvest or in multiple small applications versus a single large application. If elemental sulfur is used as a fungicide, it contributes to the plants sulfur requirement much more than affecting soil chemistry. C. E. C.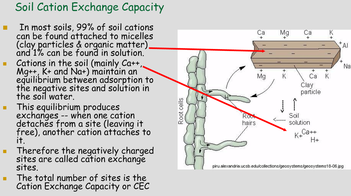 Cation Exchange Capacity is a number that represents the soils ability to hold onto and provide nutrients. A sandy soil has a C.E.C. of between 4 and 9 and cannot hold onto very many nutrients. A heavier clay soil would have a C.E.C. of over 16 and hold more nutrients than a sandy soil. The strongest nutrient holding soils have CECS in the 20’s to 30’s. CEC measures the quantity of clay and humus in a soil. By increasing the humus the CEC will increase, providing improved nutrient retention and availability. CEC goes up 2 points for every 1% increase in organic matter. Increasing a soil’s organic matter year over year by even 1% is difficult, and takes effort on the part of the farmer to replace or raise the amount of organic matter converted of lost in most crop fields annually. Cation (+) Saturation Cation saturation is the percentage of calcium, magnesium, potassium, sodium and hydrogen held on the clay and humus sites on a soil test. The ideal calcium range would be 68% to 72%. The ideal magnesium level depends on how much sand or clay the soil contains. For sandy soil the optimum range would be 16% to 20%, and for clay soils closer to 12%. The ideal potassium range would be at least 2% and 4% to 6% is better. The ideal sodium range is at least .5% and not over 3%. Formula helpful in determining the optimum nutrient levels to add.To figure desired calcium: CEC X optimum % (68%) X 400 minus existing calcium. To figure desired magnesium: CEC X optimum % (12% to 18%) X 240 minus existing magnesium. An example for calcium would be a soil with a CEC of 10.0, and a desired calcium percent of 68%. Change from PPM to #/acre on soil test by multiplying by 2. 10 X .68 X 400 = 2720 lbs. of calcium. If the soil already has 2120 lbs. of calcium, 2720-2120 = 600 lbs. of calcium needed. With 600 lbs. of calcium in a ton of limestone this soil would require a ton to raise calcium levels to 68%. CompostsOne of the best and cheapest organic fertilizers is compost. It contains organic matter, humus, calcium, phosphorus, potassium, nitrogen and many micro-nutrients, billions of microbes in each ounce and is a great food source for the biology in the soil. Compost made from plant residues and animal manures that have been fully decomposed can be applied every year at 1 to 8 tons per acre. In poor soils initial compost applications should be much higher, if the compost is fully digested and mature with a proper Carbon:Nitrogen ratio. Compost made from branches, leaves and plant residues with less manure inputs are best for hop varieties that prefer more acidic soils since this best supports fungal growth. Forests have soils that are inhabited predominantly by fungal growth. Orchard (non-tilled) soil biology closely resembles forest soil biology. Immature fresh compost containing woody residues mixed into the soil robs plants of soil nutrients (especially nitrogen). If the compost contains fresh woody residues; it is not finished and it should only be used as mulch or added to the top of the soil. Too much compost in the soil is hard for the soil to break down quickly and will temporarily tie up nutrients. If the compost has a proper C:N ratio of approx. 10-12:1, it does not tie up nutrients. High carbon composts always tie up nitrogen and sulfur, and sometimes other nutrients when worked in to the soil. Hop varieties that prefer more alkaline soils benefit from composts containing higher ratios of manure, lime and bone meal. Organic Soil Amendments – short list
Further reading and resources:
|
Details
Blog AuthorLynn, the head hop grower at Great Lakes Hops has over 30 years of experience in the horticultural field. Browse the blog articles here to find useful growing information for humulus lupulus, based on personal experience and observations at Great Lakes Hops. Archives
January 2020
Categories
All
|



 RSS Feed
RSS Feed